Stabler expression in stable cell lines

A new method relying on CRISPR/Cas9 allows the generation of stable cell lines with controlled and physiological levels of transgene expression.

A major problem with stable cell lines generated by standard methods is the heterogeneous levels of recombinant protein expression in different cell clones. This occurs because of the integration of variable numbers of the transgene or position effects caused by integration of the transgene into random locations in the genome. In addition, the use of strong heterologous promoters in transgenic vectors can drive expression to very high, non-physiological levels that harm the cells.
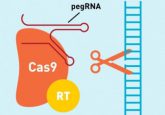
In the April issue of BioTechniques, researchers led by Brian Chen at McGill University describe an efficient method for stable cell line generation that achieves controlled, physiological expression of a transgene. Using CRISPR/Cas9 gene editing, the team inserted a single copy of a transgene at a specific gene locus, placing it under the control of an endogenous promoter with the desired level of gene expression. Their transgenic vector also includes a protein quantitation reporter gene to produce an equimolar amount of a fluorescent protein, allowing easy quantitation of the recombinant protein.