Measuring cell strength

Cells use physical force to carry out vital functions—and when that force is disrupted, disease can follow. Traditionally, measuring such force has been difficult, but a new method can now simultaneously measure cell strength in tens of thousands of cells.
Cells have the ability to generate force—shortening and re-lengthening as they carry out important functions. When that force is compromised in some way, it can lead to disease.
“Many diseases are characterized by dysregulated shortening of cells—from cancer to asthma,” said Reynold Panettieri, Jr., director of the Rutgers Institute for Translational Medicine and Science. “Any cell that shortens generates force. So if we can measure force and characterize that force, we can then look at ways to improve therapeutic approaches.”
In the past, researchers used techniques to monitor a group of four of five cells as they moved across a dish. But these methods were slow and painstaking—and couldn’t help researchers understand the more complex force patterns such as those seen in cancer metastases, where thousands of cells generate force to migrate within the body.
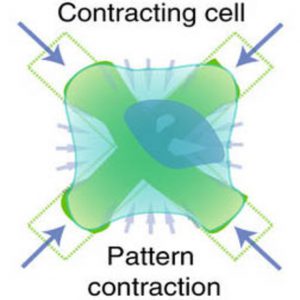
To come up with an improved method, Panettieri worked closely with bioengineering colleagues at the University of California, Los Angeles. They created a new device called fluorescently labeled elastomeric contractible surfaces (FLECS), where cells are placed on a flexible rectangular plate embedded with elastic crosses.
“Bioengineers understand very well that deformation of material is related to force. As those cells lay on the crosses and generate force, you’ll see the crosses deform,” Panettieri said.
In addition, those flexible crosses contain a molecular marker that makes them fluorescent, allowing imaging and quantification, so researchers can correlate the deformation of the cross to a certain amount of force for a single cell, or for tens of thousands of cells. The technique also offers researchers the ability to look at individual cell calcium responses and how they are related to cell strength.
Panettieri hopes this technique will elucidate the force mechanisms involved with a host of different diseases—but he said that it also could be used for drug discovery. Researchers can quickly and easily test different therapeutic molecules to see how they affect cell force, and whether they remedy any potential force issues. For example, they can simulate an asthma attack on the FLECS and then see if potential drugs can remedy it.
“We’ve now developed a very effective platform to screen hundreds of thousands of molecules to find those that will be the right drug for the right patient with the right diagnosis at the right time,” said Panettieri. “Because we can greatly shorten the length of scanning for these molecules and because of the greater fidelity we get from using precious cells from individual humans, we have the opportunity to develop precision therapies in the future.”