Copper to the rescue
Researchers accidentally discover that copper complexes inhibit the action of botulinum toxin. Could this “mistake” lead to novel therapeutic options for botulism poisoning?
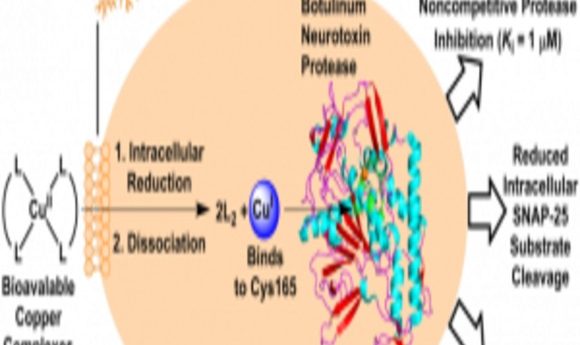
Schematic diagram of the mechanism of copper as an antidote against botulinum toxin.
Credit: Paul Bremer, The Scripps Research Institute.
In May 2017, nine people in California were hospitalized with common symptoms of drooping eyelids, slurred speech, and blurry vision, just a few days after they ate at a local gas station. Investigation revealed that the food was contaminated with a potentially fatal toxin, botulinum, produced by the bacterium, Clostridium botulinum. “It is one of the most toxic substances on the earth. It is deadly,” said Kim Janda, professor at The Scripps Research Institute, who led a recent study revealing that copper complexes might offer novel antidotes against botulinum toxin. The findings were published in the Journal of the American Chemical Society.
- botulinum enters the body through food, aerosols, or open wounds. Soon after infection, the botulinum toxin spreads via the circulatory system. It binds to nerve terminals and enters muscle cells via vesicles, where it cleaves SNAP-25, a protein essential for communication between neurons and muscles. This results in paralysis in different body parts. In severe cases, the toxin obstructs breathing by paralyzing the respiratory muscles, resulting in death.
“In general, nothing has worked [as an antidote] particularly well, partly because of the requirement for getting a really good inhibitor that interacts with the active site of this toxin. Those molecules tend not to easily get into biological membranes,” explained Michael Adler, a senior researcher at the U.S. Army Medical Research Institute of Chemical Defense, who was not involved in this study.
Janda and his team were keen on finding antidotes against the toxin. While investigating small molecules, they stumbled upon something interesting: an accidental heavy-metal contamination in their experiments inhibited the action of the toxin. This finding prodded Janda to investigate heavy metals as potential antidotes.
The team screened many different metals to look for their effects on toxin activity using a SNAP-protein-like fluorescent substrate, which fluoresces when cleaved. They noted that the fluorescence—and thus the enzyme activity of the toxin—was lowest in the presence of copper and mercury. Using crystal structure studies of the enzyme-metal complexes, the researchers determined that the metals bound to cysteine near the active site, inhibiting SNAP-25 binding.
Mercury is highly toxic to cells, so Janda’s team focused on the action of copper. They noted that organic copper complexes inhibited the cleavage of SNAP in cultured cells. Then they introduced these copper complexes into infected mice, which increased their lifespan significantly. Janda believes that these complexes release copper inside the cells, which then binds near the active site of the enzyme.
Regarding clinical trials, Adler cautioned, “If you want to take it further, you probably want to optimize its efficacy in mouse models.” Additionally, he suggested evaluating the potential toxicity of the copper complexes by testing them in other animal models. “There can be some toxic effects when using heavy metals, but for something like this where there is no antidote, it is probably not unreasonable, [to consider using them],” said Janda.