Time for a makeover, says Zika
Zika virus infection causes extensive remodeling in mammalian cells.
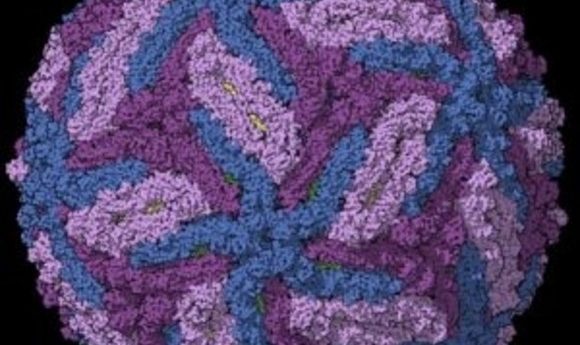
Cryo-EM derived structure of Zika virus capsid.
Credit:Sirohi, Devika, et al. “The 3.8 Å resolution cryo-EM structure of Zika virus.” Science 352.6284 (2016): 467-470.
Spring is in the air, and so are the mosquitoes that transmit the Zika virus. To curb this threat, some researchers are busy sequencing the vector genome, while others work to understand the life cycle of this emerging virus. In a new publication in Cellular Microbiology, researchers from Boston University School of Medicine show that the Zika virus extensively remodels the endoplasmic reticulum (ER) when it infects mammalian cells (1).
The team, led by Esther Bullitt, infected monkey kidney cells with Zika virus and imaged them using immunofluorescence at different time points after infection. They noticed replicating viral particles accumulating in the ER, which prodded them to take a closer look. EM images revealed major ER remodeling, including bubble-like invaginations that the researchers believe are sites of viral replication.
“They say a picture is worth a thousand words. I think in this case it’s really, really true. I mean, you can see what’s going in these cells; it’s so graphic and so obvious,” remarked Marshall Bloom from the National Institute of Allergy and Infectious Diseases, who was not involved in this study. Bloom noted that these extensive changes reinforce previous observations of Zika infection in different cell lines (2,3).
Next, Bullitt and her team investigated the fate of the virus after replication, finding that after accumulating in the ER, viral particles move one-by-one to the Golgi, where they actually become mature viruses. In the final step, the mature viruses exit the cell individually.
Bullitt suspects that following this strategy for viral replication protects the viral genetic material during replication. “Mammalian cells don’t themselves have dsRNA. The virus, in the process of copying its RNA, makes dsRNA. So, if the cell notices the presence of dsRNA, it knows it has been infected,” Bullitt explained. Thus, hiding viral dsRNA in ER membrane pockets helps the virus escape cellular defenses.
Understanding the mechanism of virus replication is of great use in therapeutics. “If you can identify a drug or a compound that can prevent the virus from [remodeling] the membrane, without at the same time killing the cell, it is sure reasonable to expect that the virus replication in the cell is going to be extremely impaired,” said Bloom.
Interestingly, the observed Zika virus life cycle is very similar to that of the Dengue virus. Bullitt and Bloom agree that this is not entirely surprising since both viruses share a common genus. Both viruses are spread by mosquitos, but Zika is also sexually transmitted and can travel from mother to fetus—processes that Bullitt is interested in exploring. Despite this mysterious difference, observed similarities in the life cycles of these viruses raise the possibility of finding common drug targets for treating both infections.