Gliding through your defenses: how parasites move
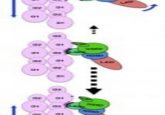
The structure and function of a key component of the ‘glideosome’ have been established, providing researchers with a better understanding of how parasites move. A recent interdisciplinary study between structural biologists and parasitologists, led by research groups from the European Molecular Biology Laboratory (Hamburg, Germany), has revealed the structure of a protein that is key to the mechanisms by which specific parasites move. This new information has shed light on how certain parasites ‘glide’ in between the cells of the host and could reveal new avenues for therapeutic development. Gliding is a behavior exhibited by parasites in the phylum Apicomplexa...
To view this content, please register now for access
Join our member community for FREE to access a collection of journal and online-only features, including:
- Exclusive access to educational videos, eBooks and insights into top BioTechniques journal articles
- The latest news and journal updates delivered straight to your inbox when you want it
- Personalized recommendations for the latest member-exclusive podcasts, interviews and expert opinions
- Priority registration to webinars, panel discussions and events
- Access to competitions and journal publication discounts, including 10% off open access fees when you sign up today!