Imaging the brain encoding memories
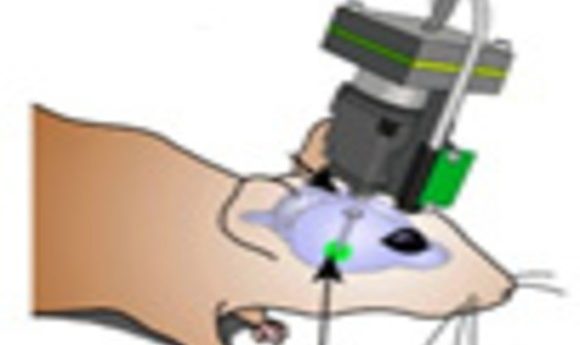
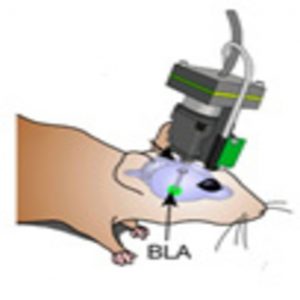
Using a miniature fluorescence microscope, researchers tracked fear learning over 6 days in mice and discovered new insights into how long-term memory is encoded in the brain.
In a classic experiment conducted by Russian physiologist Ivan Pavlov, a hungry dog was trained to salivate at the sound of a bell that was previously associated with the sight of food. This ability to associate inherently unrelated stimuli is critical for long-term memory, but it has remained unclear exactly how this process unfolds in the brain.
In a study published in Nature, researchers addressed this question by tracking the responses of thousands of neurons in the amygdala—the brain’s emotion center—in mice. “It’s been over 100 years since Pavlov did his amazing work. but we still haven’t had a glimpse of how neural ensembles encode a long-term memory,” said senior study author Mark Schnitzer of Stanford University. “This was an opportunity to examine that.”
Using a procedure called Pavlovian or classical conditioning, Schnitzer and his team repeatedly presented the conditioned stimulus (in this case, tone pulses) prior to the unconditioned stimulus (a foot shock), which caused the mice to freeze in fear. Eventually, the auditory cue alone was sufficient to elicit the freezing behavior originally evoked only by the foot shock.
“You can think of this type of learning as a survival strategy,” said Benjamin Grewe, first author of the paper. “We need that as humans; animals need that. When we associate certain stimuli with their possible dangerous outcomes, it helps us to avoid dangerous situations in the first place.”
Using a miniature fluorescence microscope, the researchers tracked the Ca2+ dynamics of ensembles of amygdala neurons in mice during fear learning over 6 days. The neural ensembles encoded associative information far more reliably than individual cells. Remarkably, the classical conditioning procedure reshaped the neural ensemble representation of the conditioned stimulus (tone pulses) to become more similar to the representation of the unconditioned stimulus (the foot shock). Moreover, the similarity of the neural ensemble representations for the auditory cue and foot shock predicted the overall extent of freezing behavior throughout learning.
“The two stimuli are both eliciting fear responses,” Schnitzer said. “It’s almost as if this part of the brain is blurring the lines between the two, in the sense that it’s using the same cells to encode them.”
Taken together, the findings support a learning model in which activation of the unconditioned stimulus representation guides the transformation of the conditioned stimulus representation. According to the authors, similar learning principles might apply to brain areas beyond the amygdala, such as other limbic regions or parts of neocortex.
In the end, the findings could have implications for studying amygdala dysfunction and the resulting emotional memory problems in patients with generalized anxiety disorder and post-traumatic stress disorder. “We’re just beginning this work,” Schnitzer said, “But these findings give us a window into how the external world may be annotated for us in this brain structure.”