How estrogen regulates bone mass
New research sheds light on exactly how estrogen helps regulate skeletal homeostasis.
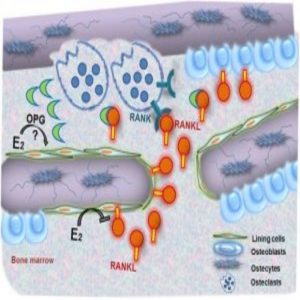
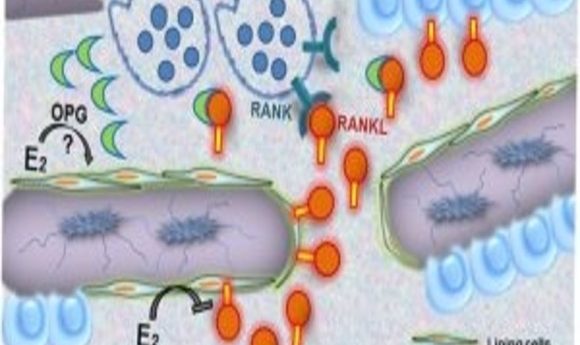
Model for how estrogen regulates osteoclastic bone resorption (1).
Estrogen is a key regulator of strong, healthy bones. Its absence can lead to diseases such as osteoporosis, where it fails to inhibit bone resorption. Researchers knew that estrogen helps regulate bone health by modulating the expression of a key cytokine, receptor activator of NF-κB ligand (RANKL), but the target cells remained unclear.
“This has been debated for many years,” said Reinhold Erben from the University of Veterinary Medicine in Vienna, Austria. “In my view, this is because of pitfalls to traditional conditional knock-out systems in mice. You cannot definitively answer the question whether this is happening in the hematopoietic or mesenchymal compartment.”
But epiphany struck from a failed experiment. A few years ago, Erben and colleagues wanted to track bone precursor cells in a host animal. They transplanted bone marrow in the hope of seeing typical tissues such as bone, muscles, and fat growing from those precursor cells.
“The funny thing is, it never worked. After weeks and months, we could never find a single donor-derived cell in these bone marrow–transplanted animals,” he explained. “But it gave us the idea that we could use this method to really separate the hematopoietic from the mesenchymal compartment. If you do it right, all the hematopoietic cells are from the donor in these bone marrow transplanted animals—but all the mesenchymal cells are host-derived. It’s a crude assay but it’s one that can help us better understand this controversy.”
The group used murine reconstitution models to selectively delete estrogen receptor-alpha (ERα) or to selectively inhibit RANKL in the two different cell-type compartments in these engineered mouse models. Then, using in situ expression profiling, they saw that cells in the mesenchymal compartment, the bone lining cells, facilitated estrogen-controlled bone resorption.
“Only if you do something to the mesenchymal cells, to the RANKL production, or to the estrogen receptor, then you see an effect on bone. It doesn’t matter if you block the RANKL coming out of the hematopoietic cell sources—that did not affect bone resorption,” he said.
Further immunohistochemistry work showed that the cells that responded most to the estrogen-deficient state in the mesenchymal compartment were the bone lining cells. “That was a surprise—we thought it must be osteocyte cells, but it wasn’t the case,” Erben explained.
Erben cautions that this work won’t immediately translate into any treatments for osteoporosis or other estrogen-related bone problems; however, “We now know more about the cells and the mechanisms of how this all works,” he said. “And this could be further exploited one day for future applications. But for now, we just gain important knowledge.”