Young at Heart: Can a Damaged Heart Muscle Repair Itself?
A new study shows that the adult mammalian heart can heal after injury.
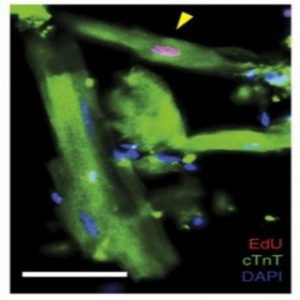
A proliferating mononuclear cardiomyocyte (yellow arrowhead) in a ventricular cell preparation from a mouse exposed to EdU after coronary artery ligation (1).
During embryonic development, mammalian heart muscle cells proliferate rapidly, but most of these cardiomyocytes quickly enter a non-proliferative state following birth. As a result, adult cardiomyocytes barely regenerate after injury, such as the damage that occurs during a heart attack, prompting researchers to look for ways to turn back the clock for these cells.
After birth, most mammalian cardiomyocytes go on to become binuclear and polyploid, but a very small population persists as mononuclear diploid cardiomyocytes (MNDCMs). Because this cell population is so small, and the regenerative capacity of the adult mammalian heart is so limited, some researchers have speculated about a relationship between MNDCMs and regeneration. Now in Nature Genetics, Henry Sucov from the University of Southern California and his colleagues reveal some details of this relationship (1).
Sucov and his team first explored the natural variability of MNDCMs across multiple strains of mice by turning to the Hybrid Mouse Diversity Panel. “This group of mice represents a broad diversity of mouse genetics,” said Sucov. The researchers surveyed MNDCM levels across 120 inbred mouse strains and found that these cells comprise 2.3%–17% of all heart cells, an unexpected degree of variation.
To look for a connection between the number of MNDCMs and the potential for regeneration following injury, the researchers surgically induced a heart attack in mice with a high (Strain A) and low (C57Bl/6) number of pre-injury MNDCMs. During recovery, the researchers exposed the mice to 5-ethynyl-2′-deoxyuridine (EdU), a marker of DNA synthesis.
The researchers found that nearly two-thirds of the EdU+ cardiomyocytes in Strain A mice were both mononuclear and diploid, indicating that these cells underwent both nuclear division and cytoplasmic division to create two new MNDCM daughter cells, evidence of a true regenerative event. When compared to the C57Bl/6 mice, the team noted a striking correlation between the starting percentage of MNDCM and the number of proliferating cells after injury.
Interested in the underlying genetic basis for the variation in MNDCM numbers between mouse strains, the researchers searched genome-wide for regions that were highly associated with variation in mononuclear cardiomyocyte frequency. One gene in particular, Tnni3k, emerged as a key player. The higher the Tnni3k expression in a particular strain, the lower the frequency of MNDCMs.
Eliminating Tnni3k expression in mice led to a significant increase in MNDCM levels. “Tnni3k is involved in some aspect of the process by which most, but not all, of those cardiomyocytes become polyploid after birth,” said Sucov. “And figuring out how it works is an ongoing area of research.”