Artificial intelligence set to revolutionize cell imaging methods
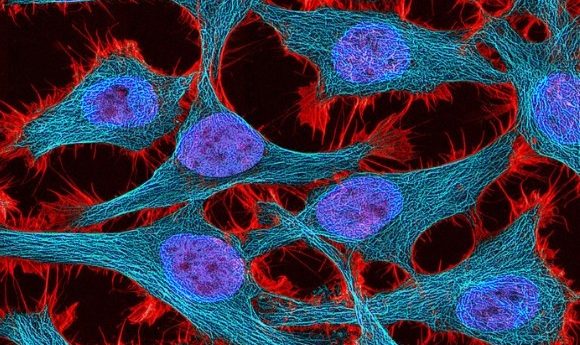
Research presented at the ASCB|EMBO 2019 meeting has highlighted how advances in computational methods have become useful tools in developing new and improved cell imaging techniques.
Understanding what is going on in cells during the progression of a disease is the first step in developing successful treatment methods. As such, there is a continuous need to develop and refine cell imaging techniques, taking advantage of new technologies as they become available.
Recently, at the 2019 joint meeting of the American Society for Cell Biology and the European Molecular Biology Organization (Washington DC, DC, USA, 7-11 December 2019), a series of abstracts were presented, each demonstrating that advances in computational methods are being utilized to develop novel techniques for cell imaging.
In the first presentation, Gil Kanfer (National Institute of Neurological Disorders and Stroke, MD, USA) described the novel method developed by his team called Artificial-Intelligence Microscopy Screening (AIMS). This technique utilizes machine learning methods to enable phenotypic-based screening of single cells at the subcellular level, allowing for the identification of pathways in mammalian cells, without the limitations of current RNAi-based screening methods.
In order to evaluate the feasibility of AIMS, the researchers screened for factors involved in TFEB translocation, into or out of the nucleus. The researchers identified both known and novel TFEB regulators, demonstrating that AIMS was successful.
Then, Anne Carpenter (Broad Institute of Harvard and MIT, MA, USA) discussed her efforts to build on an existing strategy, known as image-based profiling, using computational approaches, such as deep learning. The described technique is less expensive and provides more information than traditional methods.
- Integrating artificial intelligence and genomics
- Ge Wang and Margarida Barroso on the HOTGEM imaging technology for breast cancer
- Nano-sized laser: the future for imaging living tissues?
This approach involves staining many different cellular components and extracting thousands of morphological features from each cell’s image, using an assay known as cell painting which is then combined with artificial intelligence. Then, similarities are harvested from these profiles.
Carpenter discovered that this method could be very useful for many different applications, including diagnosing leukemia, detecting the degradation of red blood cells during blood bank storage and identifying the biological impacts of rare disease alleles.
Finally, Sangbum Park (Yale University, CT, USA) explained how combining an intravital imaging platform with a multiphoton microscope provided an opportunity to glean a deeper understanding of epidermal cells during homeostasis in live mice and revealed new principles of immune organization within the epidermis.
Despite advances in the understanding of skin homeostasis, the interaction of cells is still unclear, mostly due to an inability to simultaneously follow the behaviors of different cell types and define their functional interactions in vivo. Utilizing their technique, the researchers discovered that both populations of epidermal immune cells, the dendritic epidermal T cells and the Langerhans cells, can perceive changes to the environment and that they are able to retain spatial organization within their own population whilst the neighboring stem cells divide.
The research presented here highlighted the importance of computational technology in cell imaging techniques. These techniques could lead to a more rapid identification pathways involved in disease and consequentially accelerate the development of appropriate drug targets.