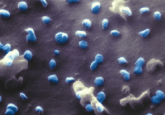
Peek behind the paper: Digital holographic microscopy – a non-invasive method to analyze the formation of spheroids
 Zahra El-Schich (left) is a researcher at Malmö University, Sweden, where she received her PhD in Medical Science. Over the years El-Schich has been interested in the technology behind digital holographic (DH) microscopy (PHIAB, Lund, Sweden) and has been successful in its use in different cancer cell biology applications. In recent years, she has investigated carbohydrates as biomarkers in cancer diagnostics, using molecularly imprinted polymers (MIPs) and advanced imaging.
Zahra El-Schich (left) is a researcher at Malmö University, Sweden, where she received her PhD in Medical Science. Over the years El-Schich has been interested in the technology behind digital holographic (DH) microscopy (PHIAB, Lund, Sweden) and has been successful in its use in different cancer cell biology applications. In recent years, she has investigated carbohydrates as biomarkers in cancer diagnostics, using molecularly imprinted polymers (MIPs) and advanced imaging.
Here, El-Schich provides an overview of her paper, recently published in BioTechniques, Digital holographic microscopy – a non-invasive method to analyze the formation of spheroids, and explains her motivations and thinking behind the study.
Please provide a short summary of your paper
DH microscopy is a unique non-invasive method for live cell analysis and monitoring. With DH microscopy, in vitro cell cultures can be imaged in both 2D and pseudo 3D, and thereafter measurements of size and morphology of the cells are provided through AppSuiteTM software (Phase Holographic Imaging AB). In this paper, a description of a novel methodology, utilizing DH microscopy for the analysis of spheroids is presented. A spheroid cell culture protocol is introduced and morphological parameters as measured by DH microscopy are presented. The study confirms the advantage of using DH microscopy for the analysis of cell spheroids by measuring spheroid morphology. In the future, if spheroids and organoids can be analyzed with DH microscopy they could be used for analyses including drug response and cell death analysis.
What inspired you to develop this new method?
I have been working with DH microscopy for many years now and initially, I could only perform my experiments on monolayers cells. In a paper published in 2015, I further developed this technique by combining DH microscopy with antibody-based microarrays so that I could successfully perform experiments also on suspension cells. During the years, I have been fascinated by the cell’s secret life. In a special moment, I captured cell apoptosis. I was interested to analyze cells’ communications with each other and sometimes you can see that cells ask for help to complete division. In this video, this is demonstrated by the cell in the middle, which tries to divide a couple of times without success.
Recently, I have been working on the EU-funded project REVERT, taRgeted thErapy for adVanced colorEctal canceR paTients. In this project, we aim to work with spheroids and organoids of colorectal cancer cells, and we are now on our way to optimizing methods and protocols for the analysis of spheroids. In this paper, I have designed a method to analyze 3D spheroids with DH microscopy.
What impact do you hope it will have on laboratory researchers?
There is a need for more methods that analyze 3D spheroids because the methods available are often limited and not compatible with 3D spheroids. I hope this method will help other researchers in this field to analyze 3D spheroids and organoids.
Do you have any tips for best practice for researchers looking to use this method?
You need first to culture your spheroids before analyzing them with DH microscopy. This part can be challenging depending on the cell types you are using. Once you improve your protocols for spheroid growth, you can start analyzing them in DH microscopy. The good thing here is that DH microscopy has automated image capture. You choose how many images to capture, and it completes the function automatically.
What are you hoping to do next in this area?
I am planning to continue using DH microscopy by treating spheroids with different drugs and monitor spheroid morphology and cell growth to improve treatment response.