Old world monkey and AIDS resistance
Using whole-genome sequencing, scientists found clues as to why some monkeys developed AIDS resistance, despite being infected with the equivalent of HIV.
Credit: Steven Bosinger, Emory School of Medicine
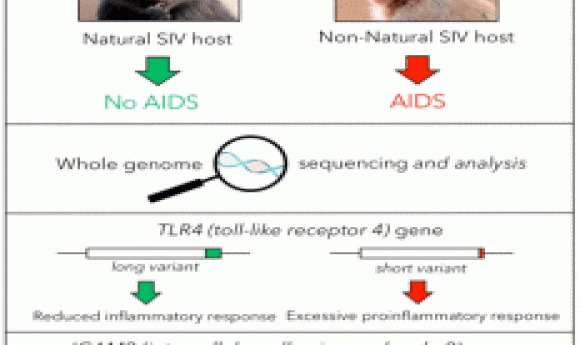
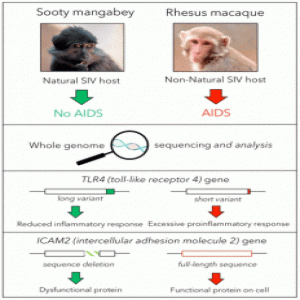
Sooty mangabeys are a species of old world monkeys that live in the forests of West Africa. They carry high viral loads of Simian Immunodeficiency Virus (SIV) in their blood, yet show no signs of illness. In a recently published study in Nature, scientists sequenced the sooty mangabey genome and compared it with the genomes of AIDS-susceptible primates to understand the reason behind AIDS resistance in this species.
“We had earlier used transcriptomic analysis to study sooty mangabeys,” said Steven Bosinger from the Emory School of Medicine, one of the leading authors of the study. “With the sequencing cost going down in the last few years, we thought it is the perfect time to study the mangabey’s genome.”
To understand the molecular mechanisms behinds the disease fighting abilities of these animals, Bosinger and his team decided to look for candidate genes in the genomes of sooty mangabeys and to compare them with those from AIDS-susceptible primates. They sequenced and assembled the genome of a sooty mangabey housed in their facility and compared it to a rhesus macaque’s genome to find the differences between the two. A total of 34 immune-related genes showed sequence diversity between mangabey and macaque. Of the divergent genes, ICAM-2 and TLR4 showed most variation in the amino acid sequence of mangabey when compared to macaque.
“We were very surprised that variations in all other immune-related genes was mild and that and TLR-4 was among the most variable,” said Bosinger.
To understand the functional implications of these mutations, Bosinger and his team studied ICAM-2 and TLR-4 in detail. They easily detected ICAM-2 in immune cells from humans and macaques but not mangabey, leading them to speculate that ICAM-2 is not functional in sooty mangabey lymphocytes. Using Western blotting, they detected a truncated form of the ICAM-2 protein in cells from mangabey.
On further investigation, the researchers found that TLR-4 protein from mangabey cells was longer than that of macaque at the C terminus by 17 amino acids. When the authors aligned the TLR-4 protein sequences from different primates, they found that this 17-amino acid long sequence is only present in AIDS-resistant hosts.
The team next performed functional studies on the mangabey TLR4 by stimulating cells with LPS, a ligand for TLR4, and then analyzing cytokine gene expression and protein production. They found reduced mRNA expression and secretion of certain pro-inflammatory cytokines in cells from mangabey compared to macaque. The researchers next linked the attenuated production of cytokines by mangabey cells with the altered TLR4 sequence.
The authors believe that future studies where the genes identified in the study are altered in vivo during SIV infection will be able to shed more light on the relationship between these genes and the disease resistance in sooty mangabey. “In the future, we would like to validate the TLR4 findings by blocking TLR4 receptors to reduce disease in macaques and see if we can develop clinically relevant therapeutics,” added Bosinger.