Not-So Identical Twins
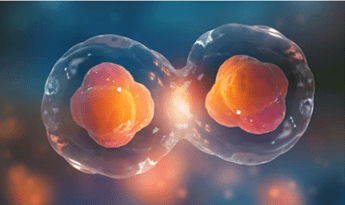
Do identical twins have the same DNA? Despite the seemingly identical DNA coding sequence, early differences in gene expression can make monozygotic twins more different than you would think. There is a common misconception that identical twins have identical DNA and, though they begin development with a matching genetic blueprint, differences in gene expression throughout life means that they differentiate more and more as they age. Though still more similar than ordinary siblings, they are no longer as identical as one may believe. These differences mean that despite their virtually identical genotype, monozygotic (MZ) twins can still be phenotypically different. With genetics often viewed as the be all and end...
To view this content, please register now for access
Join our member community for FREE to access a collection of journal and online-only features, including:
- Exclusive access to educational videos, eBooks and insights into top BioTechniques journal articles
- The latest news and journal updates delivered straight to your inbox when you want it
- Personalized recommendations for the latest member-exclusive podcasts, interviews and expert opinions
- Priority registration to webinars, panel discussions and events
- Access to competitions and journal publication discounts, including 10% off open access fees when you sign up today!