‘Mini brains’ utilized to understand brain miniaturization
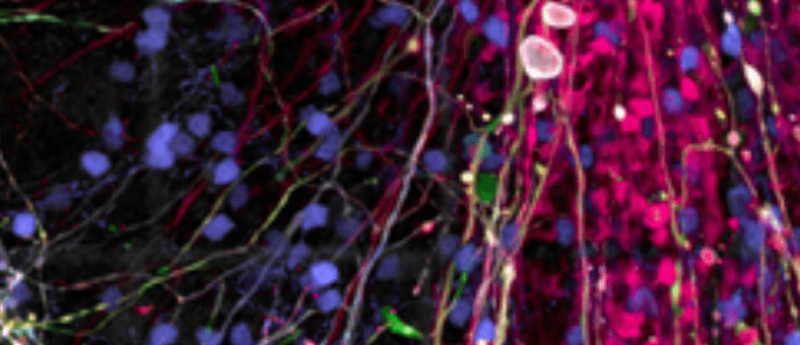
3D cerebral organoids have been utilized to model cases of cytomegalovirus-induced microcephaly.
Cytomegalovirus is a common virus that, in most cases, is harmless; however, issues can occur if infected during pregnancy. In the USA, 1 in 5 babies with congenital cytomegalovirus infection are born with birth defects or other long-term health issues, making it the most common cause of infectious-related congenital defects. Similar to the Zika virus yet a more common culprit, cytomegalovirus infection can lead to the development of microcephaly – abnormally small heads.
The basis behind cytomegalovirus-induced microcephaly has been poorly understood, largely due to difficulties in replicating the complex nature of the neurological disease in a 2D in vitro environment or in animal models. Now a team of researchers has utilized 3D organoid ‘mini brains’ to understand how infection with cytomegalovirus causes brains to shrink, or miniaturize, in utero.
The collaborative research team, led by senior author Yanhong Shi of City of Hope (CA, USA), found that when infected with a certain strain of human cytomegalovirus, the growth of the ‘mini brains’, otherwise known as cerebral organoids, was significantly reduced compared with the control models.
 Talking Techniques | Rob Vries on organoids for drug discovery and the study of COVID-19
Talking Techniques | Rob Vries on organoids for drug discovery and the study of COVID-19
BioTechniques Digital Editor, Tristan Free, speaks to Rob Vries, CEO of Hubrecht Organoid Technology (Utrecht, Netherlands), otherwise known as the HUB, to explore the HUB’s use of organoids for drug development and to further study and improve the clinical treatments of diseases such as cystic fibrosis.
Appearing to replicate what happens to babies’ brains in utero between the first and second trimester, infection with the TB40/E strain of the cytomegalovirus impaired the growth and structural development of the organoids. It also resulted in abnormal calcium signaling and defects in the functioning of the neural network.
Taking the research one step further, the team then tested a potential vaccine for cytomegalovirus-induced microcephaly in the infected organoids. A protective immune system antibody, developed in the Diamond Lab at City of Hope, was introduced to the 3D-cultured cells and it appeared that early intervention with the treatment could prevent or reduce the consequences of infection.
“Now that we have a model that replicates how [human cytomegalovirus]-induced microcephaly happens, we can use it to test antiviral agents,” commented Shi. “We can now start looking for real-world solutions.”