E. histolytica: a single-cell snapper
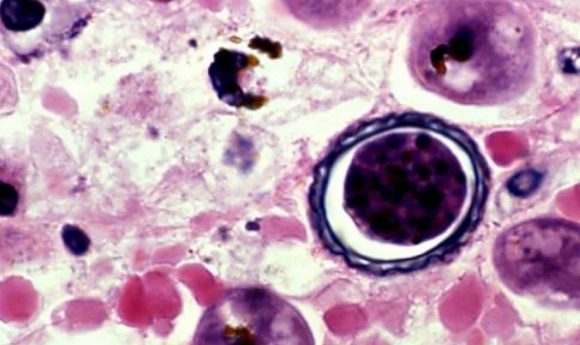
The pathogenesis of a potentially fatal amoeba, Entamoeba histolytica (E. histolytica), has been discovered, showing that the organism uses a process coined as trogocytosis, to nibble sections of a cell and present the target’s membrane proteins as an immune defense.
An infectious amoeba, E. histolytica, responsible for ulcers and potentially fatal diarrhea, has had its pathogenesis revealed by a team of researchers from the University of California, Davis (CA, USA). Led by Katherine Ralston, the team found that the organism ‘nibbles’ sections of human cells, in a process termed trogocytosis, recycling and presenting the target cell’s membrane proteins to disguise it to the host immune system.
Describing the discovery, Ralston stated that, “the amoeba quite literally takes bites out of other cells. This nibbling is how it attacks individual cells, and we think this is how it causes ulceration and damage to the human intestine.” This is a behavior that has been observed previously in regard to amoeba and immune cells, which use the process to swap sections of their cell surface.
“The amoeba quite literally takes bites out of other cells. This nibbling is how it attacks individual cells, and we think this is how it causes ulceration and damage to the human intestine.”
The research team conducted a series of experiments to test the theory that E. histolytica use trogocytosis to acquire an immunity to a host immune system, particularly the complement system of cytokines in blood serum, by appropriating membrane proteins from host cells that they can then present on their own surface.
The amoebae were exposed to human serum and their immune resistant capabilities assessed after being exposed to a situation where they were either able to conduct phagocytosis of dead cells or trogocytosis of live ones. The group that conducted trogocytosis were found to be protected while the phagocytosis group was not. Further experiments with mutant strains of E. histolytica, one strain with phagocytosis inhibited; the other with defective trogocytosis, went on to confirm this theory as the trogocytosis-enabled organisms displayed protection from the human serum’s immune response.
-
Amoebae help discover new tuberculosis drugs
-
How mold can damage the immune system
-
Do C-section babies have weaker immune systems?
The next step for this research is to identify the proteins that are targeted and presented on the amoeba’s surface, how they interact with the complement system and how they are processed by E. histolytica before presentation on the membrane. A more in-depth study of trogocytosis is also required to understand how it can be both benign and fatal to cells depending on the context of its use. “We’re really excited that this decoration with acquired proteins might apply to trogocytosis in general, because we’re realizing that it’s important in so many contexts,” exclaimed Ralston.